Nexa3D Launches Large-format Dental Lab 3D Printer
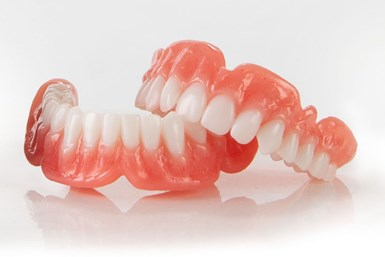
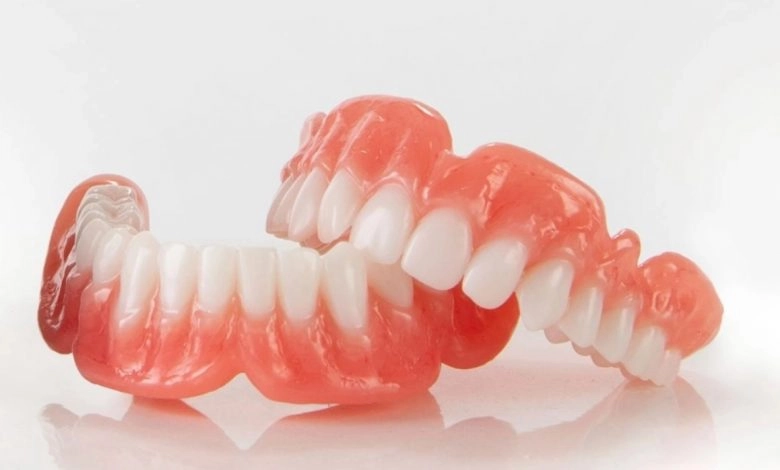
Nexa3D, launched its NXD200, a new dental lab 3D printer. NXD200 3D printer features patented LSPc technology that combines a large build area, print uniformity, advanced process algorithms, modular design for on-site automation, and intelligent optimization. The NXD200 is available with all of Keystone’s KeyPrint resins, including KeySplint Soft which delivers a combination of strength to protect teeth and flexibility for improved patient comfort, all with a transparent and aesthetic appearance.