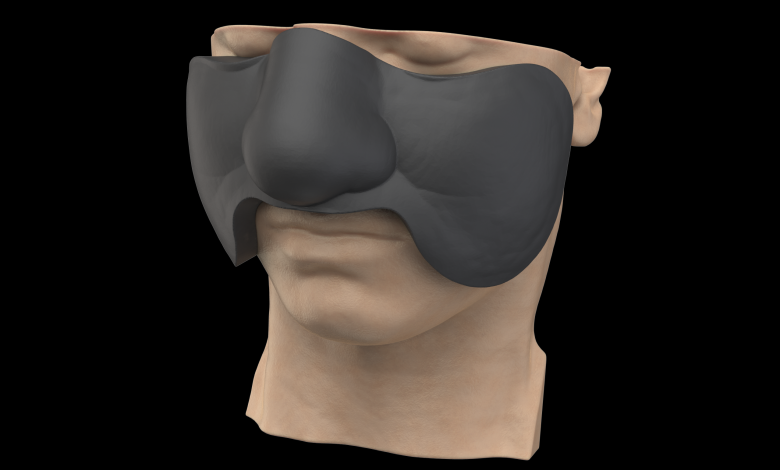
3D Systems’ VSP Bolus, a Personalized Eadiotherapy Treatment Cccessory
3D Systems has entered the radiation oncology market with FDA 510(k) clearance of VSP Bolus, a solution designed to deliver high-quality patient-specific radiotherapy treatment accessories. As the latest addition to the company’s VSP portfolio of personalized medical devices, VSP Bolus is the only solution on the market that offers a full design and production service based on the patient’s treatment plan. As a result, radiotherapy providers do not need specialized design software and expertise, allowing them to focus directly on patient treatment and care. 3D Systems’ deep experience in additive manufacturing of medical devices enables the company to deliver 3D printed, biocompatible patient-specific boluses that can improve therapy modulation as well as patient comfort.