This method, presented in the journal, was created to produce lattice scaffolds that enable precise control over its environment as well as cultured cells with particular characteristics.
“If you take cells and put them on a conventional 3D printed surface, it’s like a 2D surface to them, because the cells themselves are so much smaller,” explained Filippos Tourlomousis, a postdoctoral associate at MIT’s Center for Bits and Atoms, and first author of the research.
“But in a mesh-like structure printed using the electro-writing method, the structure is at the same size scale as the cells themselves, and so their sizes and shapes and the way they form adhesions to the material can be controlled by adjusting the porous microarchitecture of the printed lattice structure.”
3D bioprinting and melt electro writing
Tourlomousis and six others at MIT and the Stevens Institute of Technology in New Jersey sought out to accurately produce and tune cells in order to observe and control cell phenotypes; this is due to the need for tighter control over cellular function. According to the researchers, this is a major roadblock for getting tissue engineering products to the clinic.
“Any steps to tighten specifications on the scaffold, and thereby also tighten the variance in cell phenotype, are much needed by this industry,” added Tourlomousis.
“While ordinary 3D printing produces filaments as fine as 150 microns (millionths of a meter), it’s possible to get fibers down to widths of 10 microns by adding a strong electric field between the nozzle extruding the fiber and the stage on which the structure is being printed.” This method is known as melt electrowriting.
Manufacturing on Demand

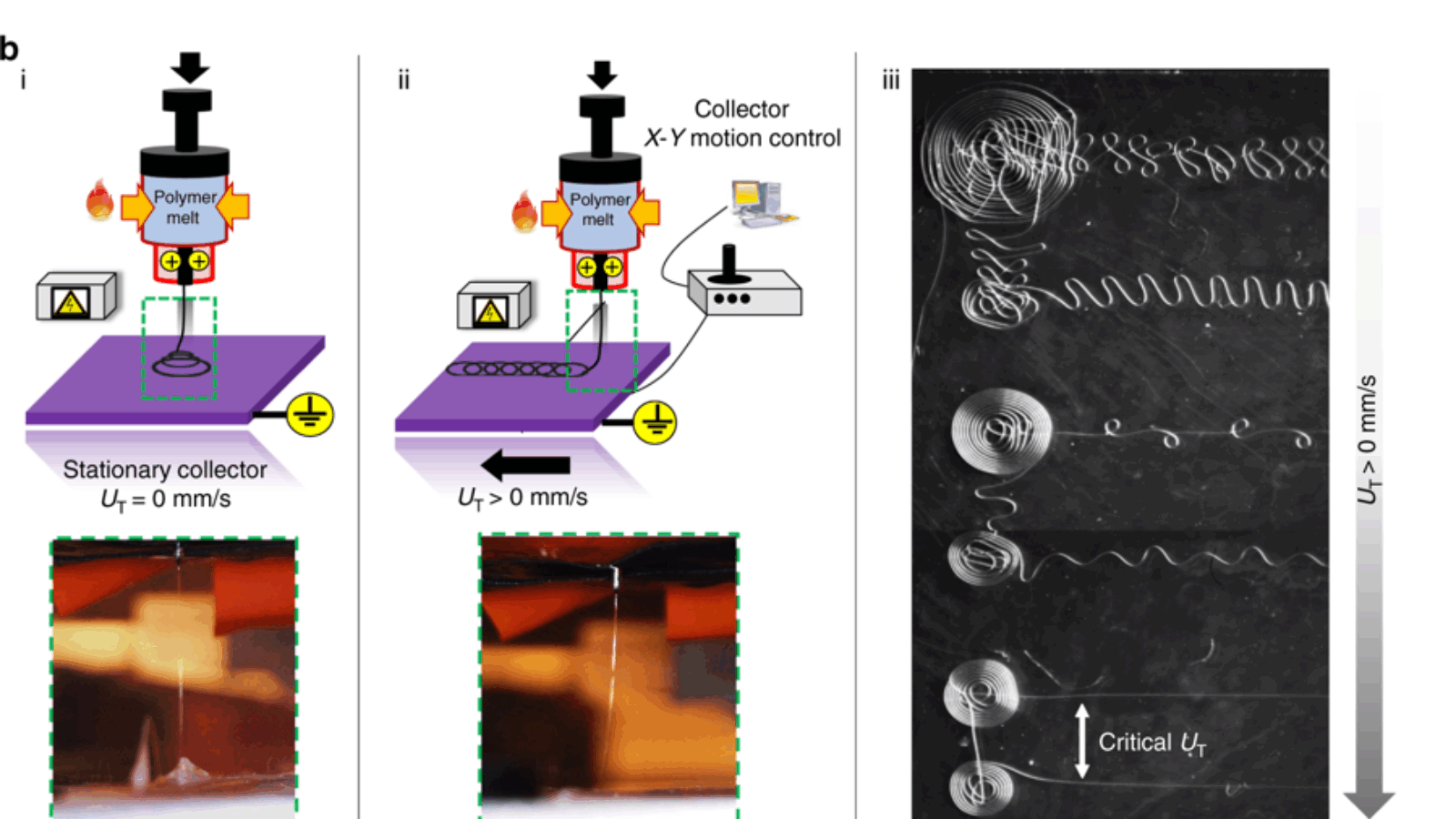
b) Direct melt electrowriting (MEW) by (i) 3D conical fiber structures are obtained by the layered deposition of fibers in circular patterns. (ii) The movement of the grounded collector plate at prescribed translational stage speeds. (iii) Micrograph depicting various fiber topographies that are obtained by tuning the translational stage speed. Image via MIT.
The study explains that cells form proteins known as focal adhesions at the places where they attach themselves to the structure. “Focal adhesions are the way the cell communicates with the external environment. These proteins have measurable features across the cell body allowing us to do metrology. We quantify these features and use them to model and classify quite precisely individual cell shapes.”
Tourlomousis stated, “It is widely known that cell shape governs cell function and this work suggests a shape-driven pathway for engineering and quantifying cell responses with great precision and with great reproducibility.”
The researchers believe this method could be used to 3D print metamaterials that can produce rare optical or electronic properties.
“Machine learning metrology of cell confinement in melt electrowritten three-dimensional biomaterial substrates” is co-authored by Thrasyvoulos Karydis, Andreas Mershin, Chao Jia, Hongjun Wang, Dilhan Kalyon, and Robert Chang.
c) 3D woven fibrous mesh with “0–90°” pore microarchitecture fabricated with direct melt electrowriting (MEW). The sample is designated MEW|0–90°. d) 3D woven fibrous mesh with “0–45°” pore microarchitecture. The sample is designated as MEW|0–45°. Image via MIT.
* This article is reprinted from 3D Printing Industry. If you are involved in infringement, please contact us to delete it.
Author: Tia Vialva


Leave A Comment