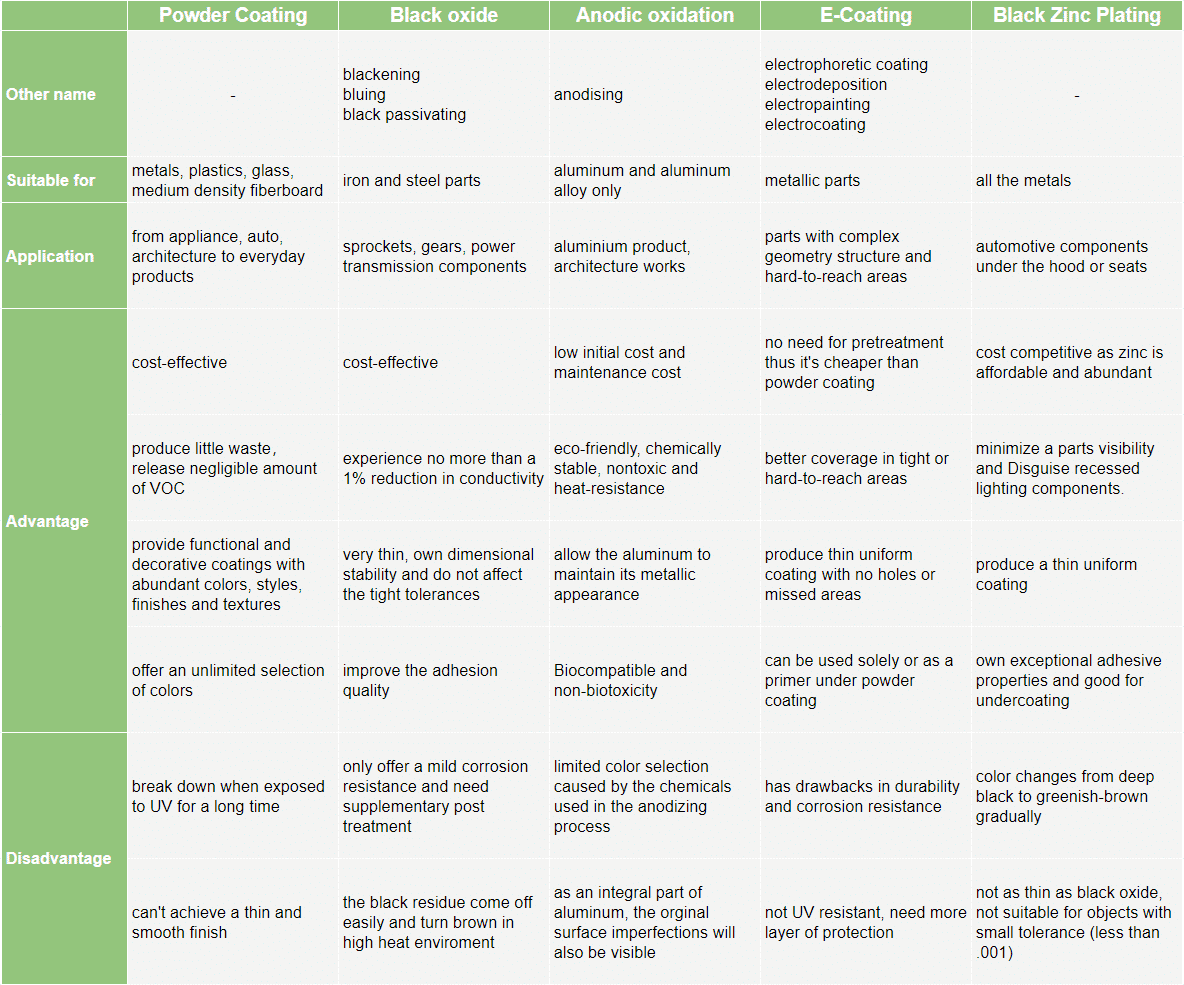
E-Coating, powder coating, black oxide, anodic oxidation, and black zinc plating are the main industrial metal coating technologies used to apply rust and corrosion-proof coating to metal parts. All of them are widely applied in the automotive and heavy machinery industries as a means of coating large components.
Despite these similarities, there are important differences to consider when selecting the correct industrial metal coating process.
What are they?
Powder Coating
While e-coating is considered to be a “wet” process, powder coating involves the application of a dry powder consisting of a precise combination of epoxy resins and various curing agents. A spray gun is used to electrostatically apply the particles onto the surface of the substrate.
The fact that the particles are electrically charged is what causes them to adhere to the surface. Curing is also used as the finishing step in the powder coating process. Curing causes the particles to melt and also serves as the catalyst for a chemical reaction that produces the desired finish.

Black oxide
Black oxide, blackening, bluing, and black passivating all refer to the process of forming a black iron oxide on the surface of ferrous metals. The black oxide coating is produced by a chemical reaction between the iron on the surface of the ferrous metal and the oxidizing salts present in the black oxide solution.
These oxidizing salts include penetrates, catalysts, activators and proprietary additives which all take part in the chemical reaction. The result of this chemical reaction is the formation of black iron oxide, magnetite (Fe3O4), on the surface of the metal being coated.

E-Coating
E-coating, also known as electrophoretic coating, electrodeposition, electropainting and electrocoating, is more similar to electroplating. With e-coating, the substrate is immersed in a bath that may consist of paint, epoxy or other water-based solution. An electric current is then used to attract the particles that are suspended in the liquid solution and deposit them onto the surface of the substrate.
The electrodeposition continues until the desired level of coating thickness is achieved, which can be regulated by increasing or decreasing the voltage level. The coated substrate is then cured in an oven to promote cross-linking.
There are two types of electrocoating: anodic and cathodic. The anodic process was invented earlier but nowadays the cathodic process is more popular. In the anodic process, negatively charged material is deposited on the positively charged component; while in the cathodic process, positively charged material is deposited on the negatively charged component.

Anodic oxidation
Anodic oxidation is an accelerated electrochemical oxidation process, which is intensified by the natural oxide skin of the metal. It involves the application of an electrical bias at relatively low currents while the substrate is immersed in an acid bath. The films can be very dense and stable, with a variety of microstructural characteristics.
Normally anodic oxidation is done in an electrochemical cell containing ~0.4 L aqueous solutions of sulfuric acid, hydrogen peroxide, and phosphoric acid at ~77°F (~25°C).

Black Zinc Plating
Black Zinc Plating refers to the color of the chromate that is applied during the post plating process. It includes a layer of zinc applied to a parts surface followed by black chromate applied over the zinc.
Black Zinc is commonly specified for its dark appearance and protective properties. Black chromate offers additional corrosion protection by protecting the zinc plating and also provides a dark to deep black appearance depending on the surface condition of the part.

The advantages of each method
Powder Coating
Powder coating protects the roughest, toughest machinery as well as the household items you depend on daily. It provides a more durable finish than liquid paints can offer, while still providing an attractive finish.
Powder coating services are suitable for various materials, including metals, plastics, glass, and medium-density fiberboard (MDF), and can provide both functional and decorative surface coatings in a wide range of colors, styles, finishes, and textures that are not easily achievable by conventional liquid coating methods.
Powder-coated products are more resistant to diminished coating quality as a result of the impact, moisture, chemicals, ultraviolet light, and other extreme weather conditions. In turn, this reduces the risk of scratches, chipping, abrasions, corrosion, fading, and other wear issues. It’s durable, eco-friendly and cheap, what a good choice!
Black oxide
Black oxide provides corrosion protection for critical-size parts. It adds no more than 5 to 10 millionths of an inch to the dimension of a part. Black oxide parts experience no more than a 1% reduction in conductivity, making it a suitable treatment for electrical parts.
The resulting lustrous black finish adds visual appeal to the parts. The harder parts tend to look glossier while the softer parts tend to look matte. It’s also cost-competitive as powder coating.
E-Coating
Because of the coating’s application, it is typically the better option when coating parts that contain hard-to-reach areas. That’s because the immersion of an object into a liquid promotes a more even and thorough distribution of the coating than can normally be achieved with a spray gun.
The powder coating application process tends to produce a thicker coating. In contrast, the e-coating process provides greater ease of regulation of the thickness level.
Anodic oxidation
Anodic oxidation of titanium allows the controlled production of a protective oxide surface layer much thicker than that formed naturally. These coatings may be dense/porous, amorphous or crystalline, depending on the conditions. Anodic oxidation is used to modify the surfaces and properties of titanium and aluminum owing to:
- Resultant mechanical properties
- Non-biotoxicity
- Biocompatible resultant mechanical properties
- Corrosion resistance
Black Zinc Plating
Black Zinc is an electrically conductive finish and offers superior corrosion protection over black oxide. Since the typical thickness is minimal, ranging from .0002” to .0006”, it is also a good alternative to powder coat or e-coat for parts with tight dimensional tolerances.
- Minimize parts visibility in a final assembled product.
- Disguise recessed lighting components.
- Common in automotive applications to camouflage components under the hood or seats.
- Cosmetic purposes requiring a uniform black finish
It is common to notice the color change from deep black to greenish-brown as the finish breaks down over time. Because of this color change, Black Zinc may not be the right finish for highly visible components with strict cosmetic requirements.
The appearance can also become compromised on parts requiring a sealer often producing streaks or a dull gray appearance, most noticeable on large flat components.

The disadvantages of each method
Black oxide
Applying a supplementary post-treatment after the black oxide finish is commonly used to increase the corrosion protection properties of black oxide coatings. Alone, black oxide offers only a very mild corrosion resistance and it can be easily rubbed off.
Powder Coating
If you are searching for a thin and smooth covering for your products, powder coating should be excluded from your alternative options. Besides, the coating tends to break down under the UV exposures. For workers, it’s less convenient to control the speed and the amount of powder coating applied.
E-Coating
Many argue that e-coating makes for even coverage with no extra buildup. However, the ultra-thin layer of deposited paint has drawbacks when it comes to durability and corrosion resistance. Further, most e-coating is not UV resistant, which means it must be followed by another layer of protection after it is baked. Often, that additional layer is applied via powder coating.
Anodic oxidation
Because of the chemicals used in the anodizing process, you are greatly limited to the color selection for your next project or product.
Anodize is an integral part of the aluminum; therefore, heavy die lines, dents, and dings on the aluminum part will show through the finish.
Black Zinc Plating
The only real downside to Black Zinc Plating is also one of it’s greatest attributes. Black Zinc is quite a bit thicker than other options which can lead to fitting issues if you are working within a very small tolerance (less than .001). In most cases, however, the added protection that is gained by the thickness is more beneficial than problematic. This is something to keep in mind but should not deter you from choosing Black Zinc.

The relationship between them
E-coating and black zinc plating both own great adhesive properties and often used as a primer before painting or other coating methods.
The powder and e-coats have no problem adhering together as long as the e-coat is adequately cured for the correct amount of time. Powder coating over e-coat provides exceptional benefits to your metal products, such as:
- Durability: When you add inner e-coating to your powder-coated products, you’ll ensure they last longer and stand up better to harsh environmental conditions. E-coating is known for its exceptional durability. It resists both outdoor elements and indoor factors like household chemicals. This outstanding performance makes it an increasingly popular choice as an added layer in powder-coated applications.
- Coverage: E-coating is the better option of the two for accessing hard-to-reach parts within your metal products. However, you might still want to add the decorative touch of powder coating. For a combination of looks and all-encompassing coverage, you can have your product e-coated, cured and then powder coated for a stylish finish.
- Corrosion and UV protection: E-coats are known for their superior corrosion resistance, which is one of the main reasons why so many companies choose the process for industrial applications. However, since most e-coats are not UV stable, they are most often used only as primers or finishes. Pairing your e-coat with a top layer of UV-resistant powder coating ensures both the corrosion protection of e-coats and the UV protection of powder coating.
It is possible to powder coat over zinc-plated steel, but it can be challenging. The plating and passivation process creates good corrosion resistance, but the surface structure is not very compatible with the powder coating process.